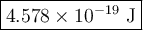Chemistry, 28.09.2019 23:10 zacksoccer8279
The blue line of the hydrogen emission spectrum has a wavelength of 433.9 nm. a hydrogen emission spectrum has a violet, a blue, a teal, and a red line. calculate the energy of one photon of this light.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 molarity koh solution, what is the molarity of the new solution
Answers: 1


Chemistry, 22.06.2019 13:50
Read the chemical equation. 2c2h2 + 5o2 → 4co2 + 2h2o which of the following statements would be correct if one mole of c2h2 was used in this reaction? one mole of oxygen was used in this reaction. five moles of oxygen were used in this reaction. four moles of carbon dioxide were produced from this reaction. two moles of carbon dioxide were produced from this reaction.
Answers: 3
You know the right answer?
The blue line of the hydrogen emission spectrum has a wavelength of 433.9 nm. a hydrogen emission sp...
Questions








Physics, 04.07.2019 18:30



Mathematics, 04.07.2019 18:30



Mathematics, 04.07.2019 18:30


Mathematics, 04.07.2019 18:30


Biology, 04.07.2019 18:30

Mathematics, 04.07.2019 18:30

History, 04.07.2019 18:30