
Chemistry, 28.09.2019 04:10 zacksoccer6937
Be sure to answer all parts. caffeine occurs naturally in coffee and tea, and is present in many soft drinks. the formula of caffeine is c, h1n402. calculate the formula mass and molar mass of caffeine. formula mass = amu molar mass = g/mol

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 18:10
The atom fluorine generally will become stable through the formation of an ionic chemical compound by accepting electron(s) from another atom. this process will fill its outer energy level of electrons.
Answers: 1

Chemistry, 22.06.2019 21:30
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1

Chemistry, 22.06.2019 22:30
What if it is did darwin used to support his theory of evolution
Answers: 1

Chemistry, 23.06.2019 00:20
4. propanol and isopropanol are isomers. this means that they have a) the same molecular formula but different chemical properties. b) different molecular formulas but the same chemical properties. c) the same molecular formula and the same chemical properties. d) the same molecular formula but represent different states of the compound
Answers: 3
You know the right answer?
Be sure to answer all parts. caffeine occurs naturally in coffee and tea, and is present in many sof...
Questions

Mathematics, 01.03.2021 22:20


Mathematics, 01.03.2021 22:20

Mathematics, 01.03.2021 22:20

Mathematics, 01.03.2021 22:20




Mathematics, 01.03.2021 22:20

History, 01.03.2021 22:20


Mathematics, 01.03.2021 22:20




Mathematics, 01.03.2021 22:20

English, 01.03.2021 22:20

Mathematics, 01.03.2021 22:20

English, 01.03.2021 22:20

Social Studies, 01.03.2021 22:20

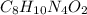
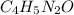
![(4\times 12)+(5\times 1)+(2\times 14)+(1\times 16)]=97amu](/tpl/images/0270/0693/7f107.png)
![(8\times 12)+(10\times 1)+(4\times 14)+(2\times 16)]=194g/mol](/tpl/images/0270/0693/8f300.png)


