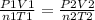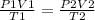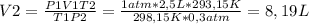Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 09:00
Scientific evidence tells us that the cause of earths four season is the tilt of earth as it revolves around the sun. the student is instructed to illustrate this information in a science notebook. how will the student illiterate winter in the northern hemisphere?
Answers: 3

Chemistry, 22.06.2019 10:40
If an area has high air pressure and low humidity, what type of weather will it most likely have? plz !
Answers: 1

Chemistry, 22.06.2019 12:30
How many grams of magnesium metal will react completely with 8.3 liters of 5.5m hcl? show all work
Answers: 1

Chemistry, 22.06.2019 14:30
How do temperature and salinity affect deepwater currents? as temperatures and salinity levels of water increase, the water rises to the surface where it creates currents as it moves to colder regions. they create changes in wind direction, moving denser water in the same direction as the wind and causing the deepwater circulation patterns found in the ocean. they equalize the forces on undersea currents caused by the coriolis effect as they replace more dense water with less dense water. they create density differences that cause dense deepwater currents to flow toward the equator where they displace less dense, warmer water above them.
Answers: 2
You know the right answer?
Aballoon filled with helium gas occupies 2.50 l at 25°c and 1.00 atm. when released, it rises to an...
Questions


Social Studies, 10.02.2021 14:00


Mathematics, 10.02.2021 14:00


Business, 10.02.2021 14:00


History, 10.02.2021 14:00


English, 10.02.2021 14:00




Physics, 10.02.2021 14:00



Health, 10.02.2021 14:00

Mathematics, 10.02.2021 14:00


Mathematics, 10.02.2021 14:00