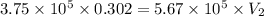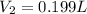Chemistry, 25.09.2019 01:20 JimmySample7
Acompressed cylinder of gas contains 45.6 mol of n2 gas at a pressure of 3.75 x 105 pa and a temperature of 23.6°c. what volume of gas has been released into the atmosphere if the final pressure in the cylinder is 5.67 x 105 pa? assume ideal behavior and that the gas temperature is unchanged.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 08:30
How does the principle of electromagnetism explain the interaction between earth’s magnetic field and the solar wind?
Answers: 1

Chemistry, 22.06.2019 13:30
In a ni-cd battery, a fully charged cell is composed of nickelic hydroxide. nickel is an element that has multiple oxidation states. assume the following proportions of the states: nickel charge proportions found 0 0.17 +2 0.3 +3 0.33 +4 0.5 (a) determine the mean of the nickel charge. enter the answer to 2 decimal places.(b) determine the cumulative distribution function of nickel charge.
Answers: 2

Chemistry, 22.06.2019 19:00
A4.86 g piece of metal was placed in a graduated cylinder containing 15.5 ml of water. the water level rose to 17.3 ml. what is the density of the metal. i need the steps of how to solve it to so i can use a formula to work out other problems.
Answers: 1

Chemistry, 22.06.2019 19:20
Consider hydrogen in an excited state n = 5n=5 that emits photons to reach the ground state. there are various possible transitions other than straight to the ground state that can occur; for example, it can drop to the n = 3n=3 state followed by the n = 3n=3 to the ground state transition. which of the possible transitions will result in the emission of a photon in the visible region?
Answers: 3
You know the right answer?
Acompressed cylinder of gas contains 45.6 mol of n2 gas at a pressure of 3.75 x 105 pa and a tempera...
Questions

Mathematics, 12.10.2020 22:01

Mathematics, 12.10.2020 22:01

Mathematics, 12.10.2020 22:01




Engineering, 12.10.2020 22:01


Mathematics, 12.10.2020 22:01


Mathematics, 12.10.2020 22:01



Chemistry, 12.10.2020 22:01

Mathematics, 12.10.2020 22:01

History, 12.10.2020 22:01

Biology, 12.10.2020 22:01


Mathematics, 12.10.2020 22:01

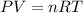
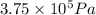 = 3675 atm (1 kPa= 0.0098 atm)
= 3675 atm (1 kPa= 0.0098 atm)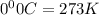
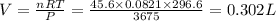
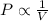 (At constant temperature and number of moles)
(At constant temperature and number of moles)
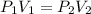
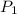 = initial pressure of gas =
= initial pressure of gas = 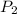 = final pressure of gas =
= final pressure of gas = 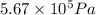
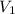 = initial volume of gas = 0.302 L
= initial volume of gas = 0.302 L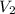 = final volume of gas = ?
= final volume of gas = ?