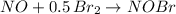Chemistry, 24.09.2019 23:20 Jordandenzel
Nitrogen monoxide, no, reacts with bromine, br2, to give nitrosyl bromide, nobr. a sample of 0.0524 mol no with 0.0262 mol br2 gives an equilibrium mixture containing 0.0311 mol nobr. what is the composition of the equilibrium mixture?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:30
Problem #3 (ch. 1, problem 15)the ideal gas law provides one way to estimate the pressure exerted by a gas on a container. the law isí‘ťí‘ť=푛푛푛푛푛푛푉푉more accurate estimates can be made with the van der waals equationí‘ťí‘ť=푛푛푛푛푛푛푉푉â’푛푛푟푟â’푞푞푛푛2푉푉2where the term nb is a correction for the volume of the molecules and the term an2/v2is a correction for molecular attractions. the values of a and b depend on the type of gas. the gas constant is r, the absolutetemperature is t, the gas volume is v, and the number of moles of gas molecules is indicated by n. if n = 1 mol of an ideal gas were confined to a volume of v = 22.41 l at a temperature of 0â°c (273.2k), it would exert a pressure of 1 atm. in these units, r = 0.0826.for chlorine gas (cl2), a = 6.49 and b = 0.0562. compare the pressure estimates given by the ideal gas law and the van der waals equation for 1 mol of cl2 in 22.41 l at 273.2 k. what is the main cause of the difference in the two pressure estimates, the molecular volume or the molecular attractions?
Answers: 1

Chemistry, 22.06.2019 03:50
Consider the reaction: n2(g) + o2(g) ? 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 09:00
Acrystal that absorvd water from air is (blank)a. aqueousb. homogenousc. hygroscopicd. efflorescent
Answers: 1

Chemistry, 22.06.2019 09:20
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2
You know the right answer?
Nitrogen monoxide, no, reacts with bromine, br2, to give nitrosyl bromide, nobr. a sample of 0.0524...
Questions

Mathematics, 27.07.2019 06:00

Physics, 27.07.2019 06:00

Mathematics, 27.07.2019 06:00



Computers and Technology, 27.07.2019 06:00





History, 27.07.2019 06:00


Chemistry, 27.07.2019 06:00


Mathematics, 27.07.2019 06:00



Biology, 27.07.2019 06:00

History, 27.07.2019 06:00

Chemistry, 27.07.2019 06:00