
Chemistry, 14.09.2019 18:10 steven0448
Using the information in the table in question 3, find the mass of a neutron and an electron relative to the mass of one proton
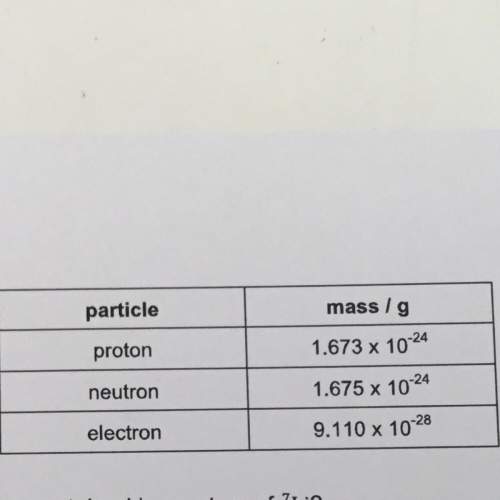

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:50
The conventional equilibrium constant expression (kc) for the system below is: 2icl(s) ⇄ i2(s) + cl2(g) [cl2] ([i2] + [cl2])/2[icl] [i2][cl2]/[icl]2 none of the listed answers are correct [i2][cl2]/2[icl]
Answers: 2


Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3

Chemistry, 22.06.2019 15:30
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 1
You know the right answer?
Using the information in the table in question 3, find the mass of a neutron and an electron relativ...
Questions

Business, 08.06.2021 14:30

Physics, 08.06.2021 14:30

Business, 08.06.2021 14:30


Spanish, 08.06.2021 14:30

Business, 08.06.2021 14:30


Mathematics, 08.06.2021 14:30

Mathematics, 08.06.2021 14:30

English, 08.06.2021 14:30

Chemistry, 08.06.2021 14:30



History, 08.06.2021 14:30

Mathematics, 08.06.2021 14:30




Mathematics, 08.06.2021 14:30

History, 08.06.2021 14:30



