
Chromium has an atomic mass of 51.9961 u and consists of four isotopes, cr50, cr52, cr53, and cr54. the cr52 isotope has a natural abundance of 83.79% and an atomic mass of 51.9405 u. the cr54 isotope has a natural abundance of 2.37% and an atomic mass of 53.9389 u. the natural abundances of the cr50 and cr53 isotopes exist in a ratio of 1: 0.4579, and the cr50 isotope has an atomic mass of 49.9460 u. determine the atomic mass of the cr53 isotope.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 19:30
The molecular formula for caffeine is cshion402. which of the following elements is not found in caffeine?
Answers: 1

Chemistry, 22.06.2019 14:20
Which of the following are sources of revenue for media companies? a. direct sales to producers b.advertising and subscriptions c. online purchase d. capital investments
Answers: 1

Chemistry, 22.06.2019 14:50
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3

Chemistry, 22.06.2019 18:00
Answer asap need it by wednesday morning carry out the following calculations on ph and ka of from data. i. calculate the ph of 0.02m hcl ii. calculate the ph of 0.036m naoh iii. calculate the ph of 0.36m ca(oh)2 iv. calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 v. calculate ka for weak acid ha which has a ph of 3.65 at 0.30m concentration vi. calculate the ka of a solution made by mixing 15.0 cm3 0.2m ha and 60.0 cm3 0.31m a-. [ph= 3.80] vii. calculate the ph of a solution made by mixing 15.0 cm3 0.1m naoh and 35.0 cm3 0.2m hcooh. [ka = 1.82 x 10-4 m]
Answers: 1
You know the right answer?
Chromium has an atomic mass of 51.9961 u and consists of four isotopes, cr50, cr52, cr53, and cr54....
Questions



Mathematics, 16.07.2019 11:20


Biology, 16.07.2019 11:20

Spanish, 16.07.2019 11:20

Social Studies, 16.07.2019 11:20


Chemistry, 16.07.2019 11:20


Chemistry, 16.07.2019 11:20



Social Studies, 16.07.2019 11:20


English, 16.07.2019 11:20

History, 16.07.2019 11:20


English, 16.07.2019 11:20

Mathematics, 16.07.2019 11:20

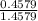 x 13.84 = 4.3469%
x 13.84 = 4.3469%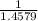 x 13.84u = 9.4931%
x 13.84u = 9.4931%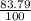 ) + ( 49.9460 x
) + ( 49.9460 x 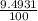 + (m₅₃ x
+ (m₅₃ x 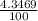 ) + (53.9389 x
) + (53.9389 x 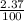 )
)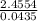 = 56.4459u
= 56.4459u

