
Rate law equation the rate of a chemical reaction depends on the concentrations of the reactants. for the general reaction between a and b, aa+bb⇌cc+dd the dependence of the reaction rate on the concentration of each reactant is given by the equation called the rate law: rate=k[a]m[b]n where k is a proportionality constant called the rate constant. the exponent m determines the reaction order with respect to a, and n determines the reaction order with respect to b. the overall reaction order equals the sum of the exponents (m+n). part a part complete what is the reaction order with respect to a? express your answer as an integer.

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 04:30
Why are people not able to scuba dive in the deep part of the ocean
Answers: 2

Chemistry, 22.06.2019 07:00
Indicate whether the specified alkyl halides will form primarily substitution products, only elimination products, both substitution and elimination products, or no products when they react with sodium methoxide. 1-bromobutane 1-bromo-2-methylpropane 2-bromobutane 2-bromo-2-methylpropane
Answers: 2

Chemistry, 22.06.2019 09:10
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2
You know the right answer?
Rate law equation the rate of a chemical reaction depends on the concentrations of the reactants. fo...
Questions






Mathematics, 18.04.2020 01:59

Mathematics, 18.04.2020 01:59

Mathematics, 18.04.2020 01:59

Geography, 18.04.2020 01:59





Biology, 18.04.2020 01:59

Mathematics, 18.04.2020 01:59





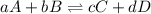
![Rate=k[A]^m[B]^n](/tpl/images/0226/8493/b2185.png)


