
Chemistry, 10.09.2019 18:30 mikayleighb2019
Why does ammonium nitrate (nh4no3) dissolve readily in water even though the dissolution process is endothermic by 26.4 kj/mol? why does ammonium nitrate (nh4no3) dissolve readily in water even though the dissolution process is endothermic by 26.4 kj/mol? the vapor pressure of the water decreases upon addition of the solute. the osmotic properties of the system lead to this behavior. the overall enthalpy of the system decreases upon addition of the solute. the overall entropy of the system increases upon dissolution of this strong electrolyte. the overall enthalpy of the system increases upon dissolution of this strong electrolyte.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:00
To save time, you can approximate the initial mass of the solid to the nearest ±1 g. for example, if you are asked to add 14.3 g of copper, add between 13 g and 15 g. which of the following sets include two samples with an equal density? which all that apply below 15.4 g gold and 18.7 g silver 15.2 g copper and 50.0 g copper 20.2 g silver and 20.2 g copper 11.2 g gold and 14.9 g gold
Answers: 1

Chemistry, 21.06.2019 23:00
An electrons position cannot be known precisely only it's probability of being in a certain location can be known
Answers: 1

Chemistry, 22.06.2019 04:00
You encounter a solution that is acidic and you decide to test it by adding a small amount of a strong acid. the ph lowers slightly but is approximately unchanged, and still remains acidic. what can you say about the solution? a. it is a buffer solution. b. it is not a buffer solution it is a strong acid solution. d. the solution has been neutralized. e. the solution has excess acid present
Answers: 1

Chemistry, 22.06.2019 05:30
Describe the interaction that occurs between two objects with the same electrical charge.
Answers: 1
You know the right answer?
Why does ammonium nitrate (nh4no3) dissolve readily in water even though the dissolution process is...
Questions



Mathematics, 28.05.2021 14:40

Physics, 28.05.2021 14:40

Geography, 28.05.2021 14:40

Mathematics, 28.05.2021 14:40

History, 28.05.2021 14:40

Chemistry, 28.05.2021 14:40

Physics, 28.05.2021 14:40


Mathematics, 28.05.2021 14:40

History, 28.05.2021 14:40


Mathematics, 28.05.2021 14:40

Engineering, 28.05.2021 14:40

Mathematics, 28.05.2021 14:40

Mathematics, 28.05.2021 14:40

Mathematics, 28.05.2021 14:40

Computers and Technology, 28.05.2021 14:40

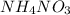 is ionic in nature. Hence, when it is added to water then it will readily dissociate into ammonium ions (
is ionic in nature. Hence, when it is added to water then it will readily dissociate into ammonium ions (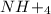 ) and nitrate ions (
) and nitrate ions (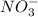 ).
).

