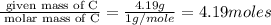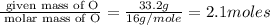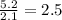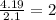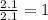Measurements show that unknown compound x has the following composition:
element mass %
...

Chemistry, 05.09.2019 17:10 samsmith666
Measurements show that unknown compound x has the following composition:
element mass %
carbon 62.4
hydrogen 4.19
oxygen 33.2
write the empirical chemical formula of x?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 13:20
An aqueous solution of calcium hydroxide is standardized by titration with a 0.120 m solution of hydrobromic acid. if 16.5 ml of base are required to neutralize 27.5 ml of the acid, what is the molarity of the calcium hydroxide solution?
Answers: 3

Chemistry, 22.06.2019 13:50
Read the chemical equation. 2c2h2 + 5o2 → 4co2 + 2h2o which of the following statements would be correct if one mole of c2h2 was used in this reaction? one mole of oxygen was used in this reaction. five moles of oxygen were used in this reaction. four moles of carbon dioxide were produced from this reaction. two moles of carbon dioxide were produced from this reaction.
Answers: 3

Chemistry, 22.06.2019 19:30
Acetylene gas c2h2 undergoes combustion to produce carbon dioxide and water vapor how many grams of water are produced by the same amount of c2h2?
Answers: 2

Chemistry, 22.06.2019 21:00
Which of the following is a physical property flammability heat of combustion solubility and toxicity
Answers: 1
You know the right answer?
Questions



History, 10.10.2019 02:40



Social Studies, 10.10.2019 02:40


Mathematics, 10.10.2019 02:40

Mathematics, 10.10.2019 02:40

Mathematics, 10.10.2019 02:40




Biology, 10.10.2019 02:40



History, 10.10.2019 02:40

Geography, 10.10.2019 02:40

Mathematics, 10.10.2019 02:40

Mathematics, 10.10.2019 02:40

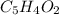 .
.