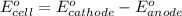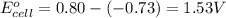Chemistry, 02.09.2019 19:20 bayleeharris8p78txa
Use the following half-reactions to construct a voltaic cell:
cr3+(aq) + 3e−\rightarrow→ cr(s) eo = −0.73 v
ag+(aq) + e−\rightarrow→ ag(s) eo = 0.80 v
determine the balanced overall redox reaction, and calculate eocell.
3 ag(s) + cr3+(aq) \rightarrow→ 3 ag+(aq) + cr(s), eocell = −1.53 v
3 ag+(aq) + cr(s) \rightarrow→ 3 ag(s) + cr3+(aq),eocell = +3.13 v
3 ag(s) + cr3+(aq) \rightarrow → 3 ag+(aq) + cr(s), eocell = −3.13 v
ag(s) + cr3+(aq) \rightarrow→ ag+(aq) + cr(s), eocell = −1.53 v
3 ag+(aq) + cr(s) \rightarrow→ 3 ag(s) + cr3+(aq), eocell = +1.53 v
ag+(aq) + cr(s) \rightarrow→ ag(s) + cr3+(aq), eocell = +1.53 v

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:20
Concerning the 10.0 ml of 0.50 m nacl to 100 ml of solution: when a solution is diluted, does it change the number of moles dissolved?
Answers: 3

Chemistry, 23.06.2019 00:00
What does an electron configuration for an atom relate to the atoms placement on the periodic table
Answers: 2

Chemistry, 23.06.2019 00:30
Fred is studying a substance that is made out of only one element. this means that
Answers: 1

Chemistry, 23.06.2019 02:00
Which best describes the present-day universe? opaque, expanding very slowly, stars produce heavy elements transparent, expanding at an accelerated rate, stars produce heavy elements opaque, expanding at an accelerated rate, stars produce only hydrogen and helium transparent, expanding very slowly, stars produce only hydrogen and helium
Answers: 1
You know the right answer?
Use the following half-reactions to construct a voltaic cell:
cr3+(aq) + 3e−\rightarrow→ cr(s...
cr3+(aq) + 3e−\rightarrow→ cr(s...
Questions

Geography, 02.09.2019 06:20

Social Studies, 02.09.2019 06:20



History, 02.09.2019 06:20

Social Studies, 02.09.2019 06:30

Chemistry, 02.09.2019 06:30

Mathematics, 02.09.2019 06:30

Mathematics, 02.09.2019 06:30

History, 02.09.2019 06:30


History, 02.09.2019 06:30

Computers and Technology, 02.09.2019 06:30


Business, 02.09.2019 06:30

Biology, 02.09.2019 06:30



Mathematics, 02.09.2019 06:30

Mathematics, 02.09.2019 06:30

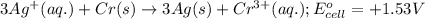
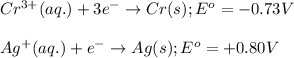
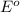 potential will always get reduced and will undergo reduction reaction. Here, silver will always undergo reduction reaction will get reduced.
potential will always get reduced and will undergo reduction reaction. Here, silver will always undergo reduction reaction will get reduced.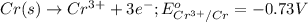
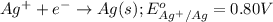 ( × 3)
( × 3)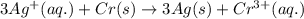
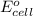 of the reaction, we use the equation:
of the reaction, we use the equation: