The following data was measured for the reaction above at a constant temperature.
experiment i...

Chemistry, 31.08.2019 05:30 4300224102
The following data was measured for the reaction above at a constant temperature.
experiment initial [a] mol l‐1 initial [b] mol l‐1 initial rate of formation of c (mol l‐1 s‐1)
1 0.10 0.10 1.12 x 10‐3
2 0.10 0.20 2.24 x 10‐3
3 0.20 0.10 4.48 x 10‐3
what is the order of the reaction with respect to a?
what is the order of the reaction with respect to b?
what is the rate law for this reaction?
calculate the rate constant.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:30
Long term exposure to waves can cause sunburns and skin cancer. a) visible b) infrared c) gamma rays d) ultraviole
Answers: 1

Chemistry, 22.06.2019 14:50
Given the following information: mass of proton = 1.00728 amu mass of neutron = 1.00866 amu mass of electron = 5.486 × 10^-4 amu speed of light = 2.9979 × 10^8 m/s calculate the nuclear binding energy (absolute value) of 3li^6. which has an atomic mass of 6.015126 amu. j/mol.
Answers: 2

Chemistry, 22.06.2019 21:00
One similarity and one difference between an element and a mixture of elements
Answers: 1

Chemistry, 23.06.2019 11:20
Which of the following is a pure substance? airbloodcopperwood
Answers: 2
You know the right answer?
Questions




Biology, 22.10.2019 01:30


Mathematics, 22.10.2019 01:30


Medicine, 22.10.2019 01:30

History, 22.10.2019 01:30



Mathematics, 22.10.2019 01:30




History, 22.10.2019 01:50

Social Studies, 22.10.2019 01:50


Social Studies, 22.10.2019 01:50

![V=k[A]^{2} [B]](/tpl/images/0214/1245/29401.png)
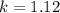
![\frac{[A]_{3}}{[A]_1{}} =2](/tpl/images/0214/1245/57061.png)
![\frac{[V]_{3}}{[V]_1{}} =4](/tpl/images/0214/1245/908c3.png)
![[A]^{2}](/tpl/images/0214/1245/66fd2.png)
![\frac{[B]_{2}}{[B]_1{}} =2](/tpl/images/0214/1245/5cc2d.png)
![\frac{[V]_{2}}{[V]_1{}} =2](/tpl/images/0214/1245/0fa6f.png)
![[B]}](/tpl/images/0214/1245/fe23b.png)
![V=k[A]^{2}[B]](/tpl/images/0214/1245/202fc.png)
![k=\frac{V}{[A]^{2}[B]}](/tpl/images/0214/1245/4243e.png)


