
Chemistry, 13.08.2019 17:10 mooreadrian4123532
What is the approximate ph at the equivalence point of a weak acid-strong base titration if 25 ml of aqueous formic acid requires 29.80 ml of 0.3567 m naoh? ka =1.8 × 10-4 for formic acid.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 14:00
How many absorptions would you expect to observe in the 13c nmr spectra of the following molecules? a) 3-chloropentane b) cis-4-methyl-2-pentene
Answers: 2

Chemistry, 22.06.2019 18:30
How many moles of lead are in 1.50 x 10^12 atoms of lead? could you explain the answer as well and not just give it to me i am refreshing for finals and i need to know how to do it
Answers: 3

Chemistry, 23.06.2019 05:00
Match each term to its description. match term definition excess reactant a) reactant that can produce a lesser amount of the product limiting reactant b) amount of product predicted to be produced by the given reactants theoretical yield c) reactant that can produce more of the product
Answers: 3

Chemistry, 23.06.2019 05:30
Stoichiometry- i need with 14 and 15! an explanation would be appreciated!
Answers: 1
You know the right answer?
What is the approximate ph at the equivalence point of a weak acid-strong base titration if 25 ml of...
Questions






Social Studies, 05.09.2019 03:20














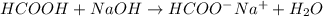
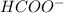
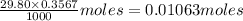
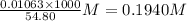
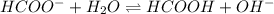
![\frac{[HCOOH][OH^{-}]}{[HCOO^{-}]}=K_{b}(HCOO^{-})=\frac{10^{-14}}{Ka(HCOOH)}](/tpl/images/0174/8862/8150e.png)
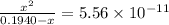
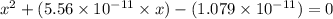
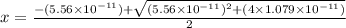
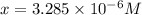
![pH=14-pOH=14+log[OH^{-}]=14+logx=14+log(3.285\times 10^{-6})=8.52](/tpl/images/0174/8862/c94a5.png)


