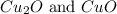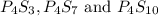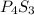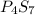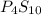Chemistry, 13.08.2019 04:20 saabrrinnaaa
The following phosphorus sulfides are known: p4s3, p4s7, and p4sio. do these compounds obey the law of multiple proportions?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 11:00
The diagram below shows the different phase transitions that occur in matter. which arrow represents the transition in which dew is formed?
Answers: 1


Chemistry, 23.06.2019 03:20
High-pressure liquid chromatography (hplc) is a method used in chemistry and biochemistry to purify chemical substances. the pressures used in this procedure range from around 500 kilopascals (500,000 pa) to about 60,000 kpa (60,000,000 pa). it is often convenient to know the pressure in torr. if an hplc procedure is running at a pressure of 1.03×108 pa , what is its running pressure in torr?
Answers: 3

Chemistry, 23.06.2019 03:30
Why do electrons further from the nucleus have more energy
Answers: 1
You know the right answer?
The following phosphorus sulfides are known: p4s3, p4s7, and p4sio. do these compounds obey the law...
Questions

Social Studies, 18.03.2021 03:10

Mathematics, 18.03.2021 03:10



Mathematics, 18.03.2021 03:10

Biology, 18.03.2021 03:10



Mathematics, 18.03.2021 03:10



Mathematics, 18.03.2021 03:10

Mathematics, 18.03.2021 03:10

Biology, 18.03.2021 03:10


Mathematics, 18.03.2021 03:10




History, 18.03.2021 03:10