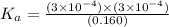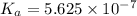Chemistry, 10.08.2019 01:20 Pauline3607
Amonoprotic weak acid, ha, dissociates in water according to the reaction: ha(aq) = h+(aq) + a−(aq). the equilibrium concentrations of the reactants and products are [ha]=0.160 m , [h+]=3.00×10^−4 m , and [a−]=3.00 ×10^−4 m. calculate the value for the acid ha .

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 15:00
Which substance is a steroid? cholesterol fatty acid monosaccharide trans fat
Answers: 1

Chemistry, 22.06.2019 16:10
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2

Chemistry, 23.06.2019 02:00
Anitrogen atom and an oxygen atom combine chemically to form nitric oxide. what is nitric oxide?
Answers: 1

Chemistry, 23.06.2019 05:00
1. true or false: minerals are inorganic. true false 2. inorganic means that something has never been found alive 3. halite is another name for and is a mineral with a cubic crystal pattern. table salt rock salt
Answers: 2
You know the right answer?
Amonoprotic weak acid, ha, dissociates in water according to the reaction: ha(aq) = h+(aq) + a−(aq)...
Questions

English, 05.11.2020 01:20

Social Studies, 05.11.2020 01:20

Biology, 05.11.2020 01:20





Mathematics, 05.11.2020 01:20

Mathematics, 05.11.2020 01:20

Chemistry, 05.11.2020 01:20

Physics, 05.11.2020 01:20



Biology, 05.11.2020 01:20

Mathematics, 05.11.2020 01:20

Chemistry, 05.11.2020 01:20

Arts, 05.11.2020 01:20



Mathematics, 05.11.2020 01:20

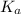 for the acid HA is,
for the acid HA is, 
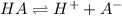
![K_a=\frac{[H^+][A^-]}{[HA]}](/tpl/images/0173/9990/66f51.png)