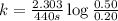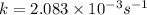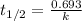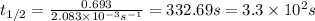Chemistry, 08.08.2019 05:30 billy583887
What is the half-life of a first-order reaction if it takes 4.4 x 102 seconds for the concentration to decrease from 0.50 m to 0.20 m? 2.5 x 102 s 3.3 x 102s 1.6s 21 s 27 s

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 16:30
10-14. (a) when 100.0 ml of weak acid ha were titrated with 0.093 81 m naoh, 27.63 ml were required to reach the equivalence point. find the molarity of ha. (b) what is the formal concentration of a- at the equivalence point? (c) the ph at the equivalence point was 10.99. find pk. for ha. (d) what was the ph when only 19.47 ml of naoh had been added?
Answers: 1

Chemistry, 22.06.2019 06:30
Identify the missing numbers below to show the result of multiplying the numbers (1.6 × 10-19)(5.0 × 106) = c × 10d
Answers: 1

Chemistry, 22.06.2019 09:10
Select the correct answer from each drop-down menu.describe what happens to a carbon-11 atom when it undergoes positron emission.the decay of a carbon-11 atom _1_, and this causes it to emit _2_.options for 1: > changes a neutron into a proton> changes a proton into a neutron> is hit with a neutron> reconfigures its protons and neutronsoptions for 2: > a negatively charged electron-sized particle> a positively charged election-sized particle> two atoms and several neutrons> two neutrons and two protons
Answers: 3

Chemistry, 22.06.2019 16:30
4. a 20-kg child is tossed up into the air by her parent. the child is 2 meters off the ground traveling 5 m/s. circle one: ke / gpe / both show your work for finding the values of each type of energy the object has:
Answers: 1
You know the right answer?
What is the half-life of a first-order reaction if it takes 4.4 x 102 seconds for the concentration...
Questions




History, 28.09.2019 13:00





Mathematics, 28.09.2019 13:00

Social Studies, 28.09.2019 13:00

Biology, 28.09.2019 13:00



History, 28.09.2019 13:00

Mathematics, 28.09.2019 13:00

Computers and Technology, 28.09.2019 13:00


Mathematics, 28.09.2019 13:00

History, 28.09.2019 13:00

Mathematics, 28.09.2019 13:00

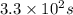
![k=\frac{2.303}{t}\log\frac{[A_o]}{[A]}](/tpl/images/0173/1549/f1041.png)
![[A_o]](/tpl/images/0173/1549/dc622.png) = initial amount of the reactant = 0.50 M
= initial amount of the reactant = 0.50 M