
Chemistry, 06.08.2019 04:20 ilovecatsomuchlolol
Which of the following describes the element kr? choose all that apply. (a) belongs to a group consisting entirely of gases forms a basic solution in water. (b) is very reactive as a metal. (c) is one of the group of the least reactive elements. (d) reacts vigorously with alkali metals to form salts. (e) consists of diatomic molecules in elemental form.

Answers: 1


Another question on Chemistry


Chemistry, 23.06.2019 06:40
The combustion of methane, ch4, releases 890.4kj/mol. that is, when one mole of methane is burned,890.4 kj are given off to the surroundings. this meansthat the products have 890.4 kj less than the reactants.thus, ah for the reaction = - 890.4 kj. a negative symbolforah indicates an exothermic reaction.ch (g) + 20 (g)> co2 (g) + 2 h0 (1); ah = - 890.4 kga) how much energy is given off when 2.00 mol of ch,are burned? b) how much energy is released when 22.4g of ch. areburned?
Answers: 1

Chemistry, 23.06.2019 07:30
Chris is about to do an experiment to measure the density of water at several temperatures. his teacher has him prepare and sign a safety contract before beginning the experiment. which term is mostlikely part of the safety contract
Answers: 3

Chemistry, 23.06.2019 12:50
Complete the paragraph to describe the characteristics of a borane molecule (bh3). the lewis structure and table of electronegativities are given. the bond polarities in bh3 are , the molecular shape is , and the molecule is .
Answers: 2
You know the right answer?
Which of the following describes the element kr? choose all that apply. (a) belongs to a group cons...
Questions

Mathematics, 20.01.2021 04:00

History, 20.01.2021 04:00


English, 20.01.2021 04:00

Mathematics, 20.01.2021 04:00



Mathematics, 20.01.2021 04:00


English, 20.01.2021 04:00

Mathematics, 20.01.2021 04:00

Mathematics, 20.01.2021 04:00

Mathematics, 20.01.2021 04:00



Mathematics, 20.01.2021 04:00


Biology, 20.01.2021 04:00


Mathematics, 20.01.2021 04:00

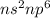 electronic configuration in their valence shell.High ionization enthalpies.Positive value of electron gain enthalpy.
electronic configuration in their valence shell.High ionization enthalpies.Positive value of electron gain enthalpy.

