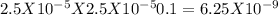Chemistry, 06.08.2019 03:20 orianabrown15
The ph of a 0.1 m mcl (m is an unknown cation) was found to be 4.7. write the net ionic equation for the hydrolysis of m and its corresponding equilibrium expression kb. calculate the value of kb

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 15:50
In a popular classroom demonstration, solid sodium is added to liquid water and reacts to produce hydrogen gas and aqueous sodium hydroxide. part a write a balanced chemical equation for this reaction. express your answer as a chemical equation. identify all of the phases in your answer.
Answers: 3

Chemistry, 21.06.2019 23:00
Write a brief passage describing a neutral atom of nitrogen-14 (n-14). describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. use the periodic table to you. 14 protons and eletrons since its a neutral atom
Answers: 1

Chemistry, 22.06.2019 07:30
What is i fracture in the crust called when land move up, down or sideways
Answers: 2

Chemistry, 22.06.2019 09:20
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2
You know the right answer?
The ph of a 0.1 m mcl (m is an unknown cation) was found to be 4.7. write the net ionic equation for...
Questions








Chemistry, 12.03.2021 15:30





Biology, 12.03.2021 15:30

Computers and Technology, 12.03.2021 15:30




English, 12.03.2021 15:30


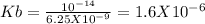

![\frac{[H^{+}][MOH]}{[M^{+}]}](/tpl/images/0172/0660/8857d.png)