
Chemistry, 31.07.2019 04:30 marieknight689
Consider the balanced chemical equation that follows. you are asked to determine how many moles of water you can form from 4 moles of hydrogen and excess oxygen. 2h2(g)+o2(g)→2h2o(l) which of the following shows calculations for a correct way to solve this problem? view available hint(s) consider the balanced chemical equation that follows. you are asked to determine how many moles of water you can form from 4 moles of hydrogen and excess oxygen. which of the following shows calculations for a correct way to solve this problem? 4 mol h2×2 mol h2o2 mol h2=4 mol h2o 4 mol h2×2 mol h2o1 mol o2=8 mol h2o 2 mol h2×2 mol h22 mol h2o=2 mol h2o

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 21:30
How air particles exert a pressure on the inside of the balloon
Answers: 1


Chemistry, 22.06.2019 14:30
The valence of aluminum is +3, and the valence of the chlorine is -1. the formula fir the aluminum chloride is correctly written as
Answers: 2

You know the right answer?
Consider the balanced chemical equation that follows. you are asked to determine how many moles of w...
Questions



Health, 09.10.2019 18:50


History, 09.10.2019 18:50

Biology, 09.10.2019 18:50


Mathematics, 09.10.2019 19:00


Mathematics, 09.10.2019 19:00


Spanish, 09.10.2019 19:00


History, 09.10.2019 19:00

History, 09.10.2019 19:00





History, 09.10.2019 19:00

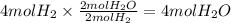
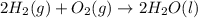
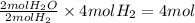 of water.
of water.

