
Chemistry, 30.07.2019 18:30 grumpygreengori
3. methane (ch4) behaves as an ideal gas under standard temperature and pressure conditions. what volume is occupied by 1 kg of methane at a temperature of 35 °c and a pressure of 0.9 atm?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 12:40
When 13.3 g koh is dissolved in 102.7 g of water in a coffee-cup calorimeter, the temperature rises from 21.4 °c to 31.53 °c. what is the enthalpy change per gram of koh (j/g) dissolved in the water? * take the density of water as 1.00 g/ml. * assume that the solution has a specific heat capacity of 4.18 j/g*k. enter to 1 decimal place. do not forget the appropriate sign /(+). canvas may auto-delete the (+) sign
Answers: 2

Chemistry, 22.06.2019 23:30
Aweight lifter raises a 1600 n barbell to a height of 2.0 meters. how much work was done? w = fd a) 30 joules b) 3000 joules c) 320 joules d) 3200 joules
Answers: 2

Chemistry, 23.06.2019 01:00
How does carbon monoxide pose the greatest threat to humans? a. it can be produced by wood fires. b. it can be produced by home furnaces. c. it is produced by acid rain. d. it is produced by modern automobiles.
Answers: 2

Chemistry, 23.06.2019 06:40
8. how much enthalpy/heat is transferred when 0.5113gof ammonia (nh3) reacts with excess oxygen according| to the following equation: 4nh3 +502 - 4n0+ 6h20ah = -905.4j
Answers: 1
You know the right answer?
3. methane (ch4) behaves as an ideal gas under standard temperature and pressure conditions. what vo...
Questions

History, 14.12.2019 08:31


Mathematics, 14.12.2019 08:31


English, 14.12.2019 08:31




Mathematics, 14.12.2019 08:31

Mathematics, 14.12.2019 08:31




Mathematics, 14.12.2019 08:31

Mathematics, 14.12.2019 08:31


Mathematics, 14.12.2019 08:31

Mathematics, 14.12.2019 09:31

Mathematics, 14.12.2019 09:31

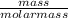
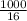 = 62.5mole
= 62.5mole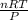
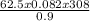 = 1711.11L
= 1711.11L


