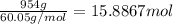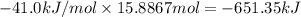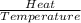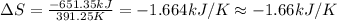Chemistry, 25.07.2019 04:20 thawkins79
The heat of vaporization δhv of acetic acid hch3co2 is 41.0 /kjmol. calculate the change in entropy δs when 954.g of acetic acid condenses at 118.1°c. be sure your answer contains a unit symbol. round your answer to 3 significant digits.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:00
Which best describes how johannes kepler developed his laws of planetary motion
Answers: 3

Chemistry, 22.06.2019 11:40
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1

Chemistry, 22.06.2019 19:00
Nan element’s square on the periodic table, the number with the greatest numerical value represents the
Answers: 3

Chemistry, 22.06.2019 23:30
Substance a is a nonpolar liquid and has only dispersion forces among its constituent particles. substance b is also a nonpolar liquid and has about the same magnitude of dispersion forces among its constituent particles. when substance a and b are combined, they spontaneously mix.
Answers: 1
You know the right answer?
The heat of vaporization δhv of acetic acid hch3co2 is 41.0 /kjmol. calculate the change in entropy...
Questions

Mathematics, 11.10.2021 20:20



English, 11.10.2021 20:20

English, 11.10.2021 20:20


Mathematics, 11.10.2021 20:20

Chemistry, 11.10.2021 20:20

English, 11.10.2021 20:20

Mathematics, 11.10.2021 20:20

Mathematics, 11.10.2021 20:20

English, 11.10.2021 20:20

Mathematics, 11.10.2021 20:20


Mathematics, 11.10.2021 20:30

Mathematics, 11.10.2021 20:30

Mathematics, 11.10.2021 20:30


Mathematics, 11.10.2021 20:30

Chemistry, 11.10.2021 20:30