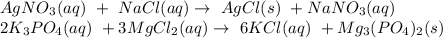Complete and balance the precipitation reactions. include physical states. refer to the solubility rules as necessary. precipitation reaction: agno_{3}(aq) + nacl(aq) -> agno3(aq)+nacl(aq)⟶ precipitation reaction: k_{3}po_{4}(aq) + mgcl_{2}(aq) -> k3po4(aq)+mgcl2(aq)⟶

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:50
Fill in the coefficients that will balance the following reaction: a0cr2(so4)3 + a1agno3 -> a2cr(no3)3 + a3ag2so4
Answers: 1



Chemistry, 23.06.2019 06:00
Which factor is likely to impact the possible number of compounds? presence of unlimited number of elements in the periodic table the inability of atoms to align perfectly with other atoms the ability of all elements to react with every other element all elements being equally reactive
Answers: 2
You know the right answer?
Complete and balance the precipitation reactions. include physical states. refer to the solubility r...
Questions









History, 17.04.2020 00:19

SAT, 17.04.2020 00:19

Mathematics, 17.04.2020 00:19

History, 17.04.2020 00:19

Mathematics, 17.04.2020 00:19

Mathematics, 17.04.2020 00:19

History, 17.04.2020 00:19

Mathematics, 17.04.2020 00:19




Mathematics, 17.04.2020 00:19