Chemistry, 13.07.2019 00:20 kashusledbetter
Understanding the high-temperature behavior of nitrogen oxides is essential for controlling pollution generated in automobile engines. the decomposition of nitric oxide (no) to n2 and o2 is second order with a rate constant of 0.0796 m−1⋅s−1 at 737∘c and 0.0815 m−1⋅s−1 at 947∘c. calculate the activation energy for the reaction in kj/mol

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:50
What happens when the temperature of a solution increases?
Answers: 2

Chemistry, 22.06.2019 18:30
How many moles of lead are in 1.50 x 10^12 atoms of lead? could you explain the answer as well and not just give it to me i am refreshing for finals and i need to know how to do it
Answers: 3

Chemistry, 23.06.2019 01:30
Which is an example of a highly unstable isotope that is often used in fission reactions?
Answers: 1

Chemistry, 23.06.2019 06:30
Consider the heating curve of h2o and line segments a, b, and c. several changes are taking place at a, b, and c. all but one would be an appropriate description as e move through segments a, b and then c.
Answers: 3
You know the right answer?
Understanding the high-temperature behavior of nitrogen oxides is essential for controlling pollutio...
Questions

Mathematics, 23.09.2021 14:00


Mathematics, 23.09.2021 14:00




Mathematics, 23.09.2021 14:00


Mathematics, 23.09.2021 14:00


Mathematics, 23.09.2021 14:00



Biology, 23.09.2021 14:00

English, 23.09.2021 14:00




Mathematics, 23.09.2021 14:00

Mathematics, 23.09.2021 14:00

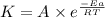
![\log (\frac{K_2}{K_1})=\frac{Ea}{2.303\times R}[\frac{1}{T_1}-\frac{1}{T_2}]](/tpl/images/0082/8135/6d953.png)
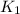 = rate constant at
= rate constant at 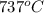 =
= 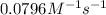
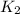 = rate constant at
= rate constant at 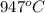 =
= 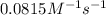
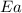 = activation energy for the reaction = ?
= activation energy for the reaction = ?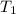 = initial temperature =
= initial temperature = 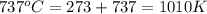
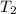 = final temperature =
= final temperature = 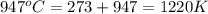
![\log (\frac{0.0815M^{-1}s^{-1}}{0.0796M^{-1}s^{-1}})=\frac{Ea}{2.303\times 8.314J/mole.K}[\frac{1}{1010K}-\frac{1}{1220K}]](/tpl/images/0082/8135/b28e1.png)