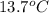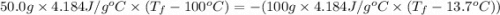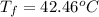Chemistry, 12.07.2019 02:30 jaylanmahone223
Calculate the final temperature of the system: a 50.0 gram sample of water initially at 100 °c and a 100 gram sample initially at 13.7 °c are mixed. the specific heat of water is 4.184 j/g°c.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 17:00
The arrangement of particles is most ordered in a sample of
Answers: 1

Chemistry, 22.06.2019 21:40
A5 mole sample of liquid acetone is converted to a gas at 75.0°c. if 628 j are required to raise the temperature of the liquid to the boiling point, 15.600 kj are required to evaporate the liquid, and 712 j are required to raise the final temperature to 75.0°c, what is the total energy required for the conversion?
Answers: 3

Chemistry, 23.06.2019 01:30
Magnesium is the limiting reactant in this experiment. calculate the theoretical yield of mgo for each trial. trial 1: trial 2: data mass of empty crucible with lid trial 1: 26.688 trial 2: 26.681 mass of mg metal, crucible, and lid trial 1: 26.994 trial: 2 26.985 mass of mgo, crucible, and lid trial 1: 27.188 trial 2: 27.180
Answers: 1

Chemistry, 23.06.2019 04:00
How many liters of water can be produced from 5.0liters of butane gas at stp, assuming excess oxygen? c4h10(g) + 02(g) → co2 (e) + h2o (g)
Answers: 2
You know the right answer?
Calculate the final temperature of the system: a 50.0 gram sample of water initially at 100 °c and...
Questions

Mathematics, 16.11.2020 20:40

Mathematics, 16.11.2020 20:40

Mathematics, 16.11.2020 20:40


Mathematics, 16.11.2020 20:40

Mathematics, 16.11.2020 20:40

English, 16.11.2020 20:40

Mathematics, 16.11.2020 20:40

Mathematics, 16.11.2020 20:40

Social Studies, 16.11.2020 20:40


Chemistry, 16.11.2020 20:40

Mathematics, 16.11.2020 20:40

Mathematics, 16.11.2020 20:40

Mathematics, 16.11.2020 20:40

Mathematics, 16.11.2020 20:40

English, 16.11.2020 20:40

Mathematics, 16.11.2020 20:40


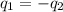
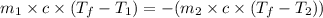
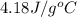
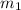 = mass of water sample with 100 °C= 50.0 g
= mass of water sample with 100 °C= 50.0 g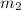 = mass of water sample with 13.7 °C= 100.0 g
= mass of water sample with 13.7 °C= 100.0 g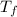 = final temperature of system
= final temperature of system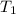 = initial temperature of 50 g of water sample=
= initial temperature of 50 g of water sample= 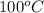
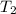 = initial temperature of 100 g of water =
= initial temperature of 100 g of water =