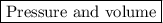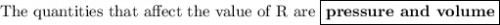Chemistry, 10.07.2019 22:20 gabbytumey
The ideal gas constant, r has several different values that could be used. which quantity causes these differences?
pressure
temperature
volume
moles
mark this and return
save and exit
next
submit

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:10
The peak wavelength for the blackbody curve of a star is in the uv range. assuming the radiation from this star can reach earth, would you be able to see it?
Answers: 2

Chemistry, 22.06.2019 23:10
Amines are good nucleophiles, even though they are neutral molecules. how would the rate of an sn2 reaction between an amine and an alkyl halide be affected if the polarity of the solvent is increased? amines are good nucleophiles, even though they are neutral molecules. how would the rate of an reaction between an amine and an alkyl halide be affected if the polarity of the solvent is increased? because both reactants in the rate-limiting step are neutral, the reaction will be faster if the polarity of the solvent is increased. because both reactants in the rate-limiting step are neutral, the reaction will be slower if the polarity of the solvent is increased. because both reactants in the rate-limiting step are neutral, the reaction will occur at the same rate if the polarity of the solvent is increased. request answer
Answers: 3

Chemistry, 23.06.2019 12:30
0.070g of hydride of carbon occupies 56cm^3 at s.t.p when vaporized and contained 14.29% by mass of hydrogen.what is the formula for the hydrocarbon
Answers: 1

Chemistry, 23.06.2019 17:30
If 2.40 moles of gas are held at a temperature of 97.0 °c in a container with a volume of 45.0 l, what is the pressure of the gas?
Answers: 1
You know the right answer?
The ideal gas constant, r has several different values that could be used. which quantity causes the...
Questions


History, 19.01.2021 20:40


Mathematics, 19.01.2021 20:40



Mathematics, 19.01.2021 20:40

Social Studies, 19.01.2021 20:40


History, 19.01.2021 20:40

Mathematics, 19.01.2021 20:40

Business, 19.01.2021 20:40

Chemistry, 19.01.2021 20:40


History, 19.01.2021 20:40

Mathematics, 19.01.2021 20:40



Mathematics, 19.01.2021 20:40

Mathematics, 19.01.2021 20:40