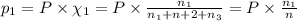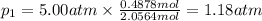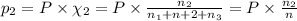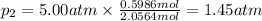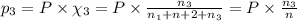Chemistry, 08.07.2019 21:10 cschellfamily
Three gases (8.00 g of methane, ch4, 18.0 g of ethane, c2h6, and an unknown amount of propane, c3h8) were added to the same 10.0-l container. at 23.0 ∘c, the total pressure in the container is 5.00 atm . calculate the partial pressure of each gas in the container.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:30
Summarize possible ways in which phases of matter could combine to form a solution.
Answers: 2

Chemistry, 22.06.2019 21:30
Harry lives in a city, and he has a lung condition known as asthma. on certain days, harry has to stay inside because pollutants in the air make it difficult for him to breathe. which of these pollution sources are nonpoint sources that might bother harry if he goes outside? choose the two that apply.
Answers: 3

Chemistry, 22.06.2019 22:30
How do limiting factors most affect population size? ostop population growthrestrict population growthincrease population sizeresult in positive impactso
Answers: 1

Chemistry, 23.06.2019 02:00
Calculate the molarity of each aqueous solution: a. 78.0 ml of 0.240 m naoh diluted to 0.250 l with water b. 38.5 ml of 1.2 m hno3 diluted to 0.130 l with water
Answers: 1
You know the right answer?
Three gases (8.00 g of methane, ch4, 18.0 g of ethane, c2h6, and an unknown amount of propane, c3h8)...
Questions


Biology, 18.07.2019 20:00


History, 18.07.2019 20:00

Mathematics, 18.07.2019 20:00





Business, 18.07.2019 20:00


History, 18.07.2019 20:00

Biology, 18.07.2019 20:00


Social Studies, 18.07.2019 20:00





Social Studies, 18.07.2019 20:00

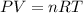 (Ideal gas equation)
(Ideal gas equation)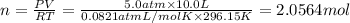
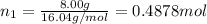
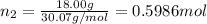
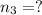
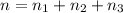

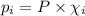
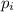 = partial pressure of 'i' component.
= partial pressure of 'i' component.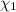 = mole fraction of 'i' component in mixture
= mole fraction of 'i' component in mixture