
Chemistry, 05.02.2020 13:46 bettinger6525
In the haber reaction, patented by german chemist fritz haber in 1908, dinitrogen gas combines with dihydrogen gas to produce gaseous ammonia. this reaction is now the first step taken to make most of the world's fertilizer.
suppose a chemical engineer studying a new catalyst for the haber reaction finds that 505 liters per second of dinitrogen are consumed when the reaction is run at 172 oc and 0.88 atm. calculate the rate at which ammonia is being produced. give your answer in kilograms per second. be sure your answer has the correct number of significant digits.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 08:40
What is the value of keq for the reaction expressed in scientific notation?
Answers: 1

Chemistry, 22.06.2019 11:00
Imagine that twenty i.u.’s of enzyme z were catalyzing the above reaction for one minute, under vmaxconditions, in a 3.00 ml assay volume. the assay is buffered with 20 mm phosphate buffer, ph 7.60. what will the ph be at the end of that one minute?
Answers: 2


Chemistry, 22.06.2019 18:30
How many moles of lead are in 1.50 x 10^12 atoms of lead? could you explain the answer as well and not just give it to me i am refreshing for finals and i need to know how to do it
Answers: 3
You know the right answer?
In the haber reaction, patented by german chemist fritz haber in 1908, dinitrogen gas combines with...
Questions

Advanced Placement (AP), 21.07.2019 16:00

Social Studies, 21.07.2019 16:00


Mathematics, 21.07.2019 16:00


English, 21.07.2019 16:00

History, 21.07.2019 16:00


Mathematics, 21.07.2019 16:00


Chemistry, 21.07.2019 16:00

Mathematics, 21.07.2019 16:00


History, 21.07.2019 16:00

English, 21.07.2019 16:00

Mathematics, 21.07.2019 16:00



Social Studies, 21.07.2019 16:00

Mathematics, 21.07.2019 16:00

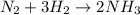 Haber reaction
Haber reaction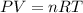
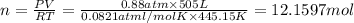
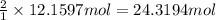 of ammonia
of ammonia

