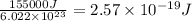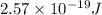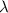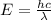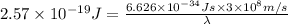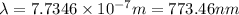Chemistry, 06.07.2019 00:30 Person51761
It takes 155. kj/mol to break a fluorine-fluorine single bond. calculate the maximum wavelength of light for which a flouine-flouring single bond could be broken by absorbing a single photon

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 21:40
During trial 2, what allowed you to determine that aluminum was the limiting reactant? check all that apply. all of the copper dissolved. all of the aluminum dissolved. the solution turned clear. the number of grams of copper(ii) chloride used in the reaction was greater than the number of grams of aluminum. the molar ratio of copper(ii) chloride to aluminum was greater than 3: 2, the equation’s molar ratio.
Answers: 2

Chemistry, 22.06.2019 09:30
Which ocean zone has the most abundant primary producer and why a) the abyssopelagic zone ,du to the absence of light and cold water temperatureb) the bathypelagic zone, due to the absence of light and cold water temperaturec) the mesopelagic zone ,due to uts high light availability and warm water temperature d) the epipelagic zone,due to its high light availability and warm water temperature
Answers: 3

Chemistry, 22.06.2019 10:50
How many grams of oxygen gas are contained in a 15 l sample at 1.02 atm and 28°c? show your work.
Answers: 1

Chemistry, 22.06.2019 13:30
Some animals that try to adapt to climate changes eventually die due to starvation, as climate change alters the web.
Answers: 2
You know the right answer?
It takes 155. kj/mol to break a fluorine-fluorine single bond. calculate the maximum wavelength of l...
Questions

Mathematics, 03.05.2021 21:50

Mathematics, 03.05.2021 21:50

Mathematics, 03.05.2021 21:50

Business, 03.05.2021 21:50


Mathematics, 03.05.2021 21:50






Mathematics, 03.05.2021 21:50




History, 03.05.2021 21:50


Chemistry, 03.05.2021 21:50

Advanced Placement (AP), 03.05.2021 21:50

Physics, 03.05.2021 21:50

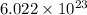 molecules = 155000 J
molecules = 155000 J