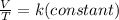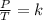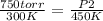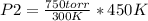Chemistry, 27.06.2019 23:40 luisannavasquez6129
If you doubled the volume of a sample of gas and
then doubled the volume again without changing
its pressure, how would the absolute temperature
of the gas be affected?
a
what pressure must be applied to keep a gas at
750 torr and 300 k at a constant volume of 200
ml if the temperature is raised to 450 k?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:50
Which of the following statements about acidic water is true? a. acid has no effect on the h,o molecules. b. the solution contains a larger number of oh ions than h,o ions. c. the solution contains a larger number of h,o ions than qh ions. d. the solution contains an equal number of h,o ions and oh ions. none of the above e.
Answers: 1

Chemistry, 22.06.2019 10:20
Gwhich r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? which r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? −ch2−oh −ch2−o||c−nh2 −ch2−coo− −ch2−ch2−ch2−ch2−n+h3
Answers: 3

Chemistry, 22.06.2019 11:30
What is the main reason why some developing countries fear the increase the free trade policies around the world?
Answers: 2

Chemistry, 22.06.2019 12:10
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3
You know the right answer?
If you doubled the volume of a sample of gas and
then doubled the volume again without changing...
then doubled the volume again without changing...
Questions

Biology, 05.12.2019 19:31

Health, 05.12.2019 19:31



Mathematics, 05.12.2019 19:31




Mathematics, 05.12.2019 19:31



History, 05.12.2019 19:31


Mathematics, 05.12.2019 19:31





Mathematics, 05.12.2019 19:31