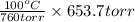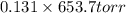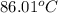Chemistry, 28.01.2020 01:31 annaclaire22
How do you calculate the boiling point of pure water at 653.7 torr?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 13:50
What happens when an atom of sulfur combines with two atoms of chlorine to produce sci2? a. each chlorine atom shares a pair of electrons with the sulfur atom. b. an electron is transferred from each chlorine atom to the sulfur atom. c. an electron is transferred from the sulfur atom to each chlorine atom. d. each chlorine atom shares all its valence electrons with the sulfur atom.
Answers: 2

Chemistry, 22.06.2019 20:00
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3

Chemistry, 23.06.2019 00:30
If there are 3.5 moles of koh, how many moles of naoh can be produced? question 1 options: a)3.0 moles naoh b)3.5 moles naoh c)1 moles naoh d)9 moles naoh
Answers: 1

Chemistry, 23.06.2019 05:00
1. true or false: minerals are inorganic. true false 2. inorganic means that something has never been found alive 3. halite is another name for and is a mineral with a cubic crystal pattern. table salt rock salt
Answers: 2
You know the right answer?
How do you calculate the boiling point of pure water at 653.7 torr?...
Questions

Biology, 15.01.2021 21:30



Mathematics, 15.01.2021 21:30


Chemistry, 15.01.2021 21:30

Mathematics, 15.01.2021 21:30

Health, 15.01.2021 21:30


Mathematics, 15.01.2021 21:30

Mathematics, 15.01.2021 21:30

Mathematics, 15.01.2021 21:30




Mathematics, 15.01.2021 21:40


Mathematics, 15.01.2021 21:40

History, 15.01.2021 21:40

Mathematics, 15.01.2021 21:40