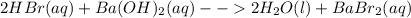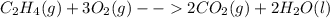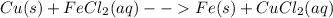Classify each of these reactions. 2hbr(aq)+ba(oh)2(aq)⟶2h2o(l)+babr2( aq) 2hbr(aq)+ba(oh)2(aq)⟶2h2o(l)+babr2( aq) c2h4(g)+3o2(g)⟶2co2(g)+2h2o(l) c2h4(g)+3o2(g)⟶2co2(g)+2h2o(l) cu(s)+fecl2(aq)⟶fe(s)+cucl2(aq) cu(s)+fecl2(aq)⟶fe(s)+cucl2(aq) na2s(aq)+2agno3(aq)⟶2nano3(aq)+ag2s (s) na2s(aq)+2agno3(aq)⟶2nano3(aq)+ag2s (s)

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:00
Write the empirical chemical formula of calcium with a mass percent of 38.8, phosphorus with a mass percent of 20.0, and oxygen with a mass percent of 41.3.
Answers: 1

Chemistry, 22.06.2019 06:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 3

Chemistry, 22.06.2019 06:30
Particle model to predict what will happen if a sharp object creates a hole in the soccer ball
Answers: 2

Chemistry, 22.06.2019 13:10
Select the correct answer a modure consists of glucose and water. what is the percent composition of glucose in the mixture if it contains 1.3 moles of glucose (cho total mass of the mature is 276 grams? ) and the a 1775
Answers: 1
You know the right answer?
Classify each of these reactions. 2hbr(aq)+ba(oh)2(aq)⟶2h2o(l)+babr2( aq) 2hbr(aq)+ba(oh)2(aq)⟶2h2o(...
Questions

Mathematics, 09.12.2020 04:00


History, 09.12.2020 04:00




Mathematics, 09.12.2020 04:00

Chemistry, 09.12.2020 04:00

Biology, 09.12.2020 04:00


Mathematics, 09.12.2020 04:00



Mathematics, 09.12.2020 04:00





Mathematics, 09.12.2020 04:00