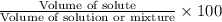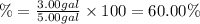Chemistry, 14.11.2019 23:31 adriandehoyos1p3hpwc
Automobile antifreeze is a mixture that consists mostly of water and ethylene glycol (c2h4(oh)2). what is the volume percent the ethylene glycol in the antifreeze mixture if it is created by mixing 3.00 gal of ethylene glycol with 2.00 gal water?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 12:20
Adeuteron, 21h, is the nucleus of a hydrogen isotope and consists of one proton and one neutron. the plasma of deuterons in a nuclear fusion reactor must be heated to about 3.02×108 k . what is the rms speed of the deuterons? express your answer using two significant figures.
Answers: 1

Chemistry, 22.06.2019 13:00
12. calculate the hydroxide ion concentration of a solution with ph = 3.25. show all calculations leading to an answer
Answers: 3

Chemistry, 22.06.2019 19:40
What type of electromagnetic waves does the human eye see as the colors red blue or green a visible light waves b radio waves c infrared waves d microwaves
Answers: 1

Chemistry, 22.06.2019 22:30
Amedication is given at a dosage of 3.000 mg of medication per kg of body weight. if 0.1500 g of medication is given, then what was the patient's weight in pounds (lbs)? there are 453.59g in 1 lb.
Answers: 2
You know the right answer?
Automobile antifreeze is a mixture that consists mostly of water and ethylene glycol (c2h4(oh)2). wh...
Questions

Mathematics, 27.10.2020 01:00

Biology, 27.10.2020 01:00

Social Studies, 27.10.2020 01:00


Engineering, 27.10.2020 01:00

Health, 27.10.2020 01:00

Mathematics, 27.10.2020 01:00

Geography, 27.10.2020 01:00



Mathematics, 27.10.2020 01:00

Mathematics, 27.10.2020 01:00


Physics, 27.10.2020 01:00

Mathematics, 27.10.2020 01:00

Mathematics, 27.10.2020 01:00

English, 27.10.2020 01:00


Mathematics, 27.10.2020 01:00

Mathematics, 27.10.2020 01:00