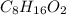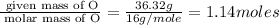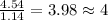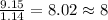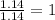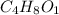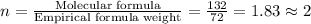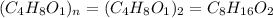Chemistry, 21.10.2019 16:50 arnold2619
Acompound is 54.53% c,54.53% c, 9.15% h,9.15% h, and 36.32% o36.32% o by mass. what is its empirical formula? insert subscripts as needed. empirical formula: chocho the molecular mass of the compound is 132 amu.132 amu. what is its molecular formula? insert subscripts as needed. molecular formula: cho

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:00
What stress will shift the following equilibrium system to the left? n2(g) + 3h2(g) ⇌ 2nh3(g) adding more n2(g) adding more nh3(g) increasing the pressure of the system reducing the volume of the container
Answers: 1

Chemistry, 22.06.2019 05:00
Choose all the answers that apply. ionic compounds dissolve easily in water do not dissolve in water have low melting points have high melting points conduct electricity when melted
Answers: 1

Chemistry, 22.06.2019 15:00
Large helium-filled balloons are used to lift scientific equipment to high altitudes. what is the pressure inside such a balloon if it starts out at sea level with a temperature of 10.0ºc and rises to an altitude where its volume is twenty times the original volume and its temperature is – 50.0ºc ?
Answers: 2

Chemistry, 22.06.2019 19:30
Chlorine and water react to form hydrogen chloride and oxygen, like this: 2cl2 (g) + 2h2o (g) → 4hcl (g) + o2 (g) also, a chemist finds that at a certain temperature the equilibrium mixture of chlorine, water, hydrogen chloride, and oxygen has the following composition: compound concentration at equilibrium cl2 0.55m h2o 0.57m hcl 0.53m o2 0.34m calculate the value of the equilibrium constant kc for this reaction. round your answer to 2 significant digits.
Answers: 2
You know the right answer?
Acompound is 54.53% c,54.53% c, 9.15% h,9.15% h, and 36.32% o36.32% o by mass. what is its empirical...
Questions

Mathematics, 15.04.2020 23:18






Business, 15.04.2020 23:18


History, 15.04.2020 23:18

Mathematics, 15.04.2020 23:18



Mathematics, 15.04.2020 23:18


Mathematics, 15.04.2020 23:18





Mathematics, 15.04.2020 23:19