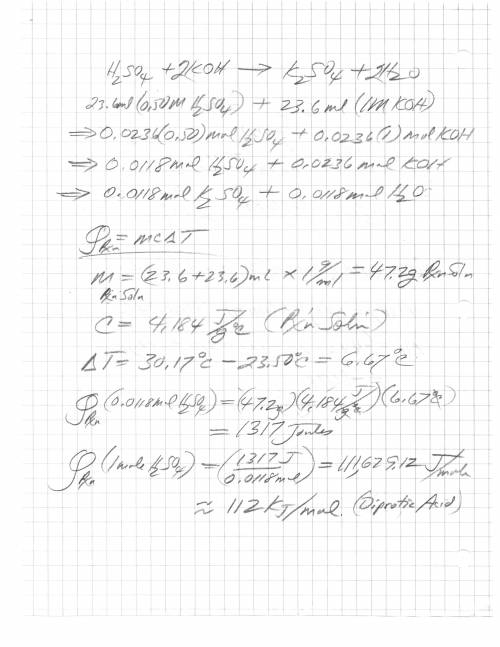Chemistry, 03.02.2020 01:53 wwesuplexcity28
When 23.6 ml of 0.500 m h2so4 is added to 23.6 ml of 1.00 m koh in a coffee-cup calorimeter at 23.50°c, the temperature rises to 30.17°c. calculate δh of this reaction. (assume that the total volume is the sum of the individual volumes and that the density and specific heat capacity of the solution are the same as for pure water.) (d for water = 1.00 g/ml; c for water = 4.184 j/g·°c.)

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 10:30
Astudent reacts 13 moles of iron with 21 moles of oxygen according to the following equation:
Answers: 2

Chemistry, 22.06.2019 12:30
Avariable that is not being directly tested during an experiment should be
Answers: 1

Chemistry, 22.06.2019 14:10
13. a covalent bond between two atoms is likely to be polar if: a. one of the atoms is much more electronegative than the other. b. the two atoms are equally electronegative. c. the two atoms are of the same element. d. the bond is part of a tetrahedrally shaped molecule. e. one atom is an anion.
Answers: 1
You know the right answer?
When 23.6 ml of 0.500 m h2so4 is added to 23.6 ml of 1.00 m koh in a coffee-cup calorimeter at 23.50...
Questions


History, 24.06.2019 12:30


Chemistry, 24.06.2019 12:30

Mathematics, 24.06.2019 12:30


Mathematics, 24.06.2019 12:30

English, 24.06.2019 12:30

Mathematics, 24.06.2019 12:30



History, 24.06.2019 12:30

Mathematics, 24.06.2019 12:30

History, 24.06.2019 12:30


History, 24.06.2019 12:30



Biology, 24.06.2019 12:30