
Chemistry, 21.06.2019 16:20 maevemboucher78
Aluminum reacts with chlorine gas to form aluminum chloride via the following reaction: 2al(s)+3cl2(g)→2alcl3(s) what is the maximum mass of aluminum chloride that can be formed when reacting 32.0 g of aluminum with 37.0 g of chlorine? express your answer to three significant figures and include the appropriate units.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 20:10
Suppose you mix one mole of sulfuric acid (h2so4) with 1 mole of sodium hydroxide(naoh). why does the ph of the solution remain below 7? ( explain so i can get better understanding! )
Answers: 2

Chemistry, 23.06.2019 02:00
What is the source of continuous heat and energy that we receive from the sun
Answers: 2

Chemistry, 23.06.2019 02:30
Asubstance is held in an open container. its particles move past one another at random speeds but do not leave the container. heat is removed from the system, and the particles slow down. when enough heat is removed, the particles no longer have enough speed to overcome the weak attractive forces between them. when this happens, the substance enters its solid state. the process described above is known as .
Answers: 3

Chemistry, 23.06.2019 03:00
You have a sample of a metal, the sample is exactly 6.02 x 1023atom, if the sample has a mass 55.85 what metal is your sample made of?
Answers: 2
You know the right answer?
Aluminum reacts with chlorine gas to form aluminum chloride via the following reaction: 2al(s)+3cl2...
Questions

Advanced Placement (AP), 25.01.2021 20:50



Business, 25.01.2021 20:50

Biology, 25.01.2021 20:50

Mathematics, 25.01.2021 20:50


Arts, 25.01.2021 20:50

Mathematics, 25.01.2021 20:50


English, 25.01.2021 20:50



Mathematics, 25.01.2021 20:50


Mathematics, 25.01.2021 20:50


Mathematics, 25.01.2021 20:50

Mathematics, 25.01.2021 20:50

English, 25.01.2021 20:50

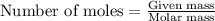 ....(1)
....(1) 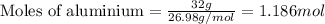
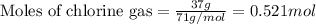
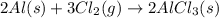
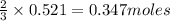 of aluminium.
of aluminium.


