
Chemistry, 22.06.2019 15:30 lovebaeforlife351
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins. co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:10
Stage in which a typical star has completely stopped fusion
Answers: 1


Chemistry, 22.06.2019 19:30
What is the common name for the compound shown here? enter the common name of the compound shown?
Answers: 2

Chemistry, 22.06.2019 21:20
40dm3 of gas at 760 torr are heated from 5°c to 50°c what is the new volume
Answers: 3
You know the right answer?
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as...
Questions


History, 29.06.2019 13:50




History, 29.06.2019 13:50






Mathematics, 29.06.2019 13:50



Mathematics, 29.06.2019 13:50

Arts, 29.06.2019 13:50


Mathematics, 29.06.2019 13:50


English, 29.06.2019 13:50

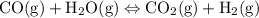 when the volume is decreased then the pressure will increase then the equilibrium will shift towards the fewer and then more number of the gas moles and hence, will have no net effect.
when the volume is decreased then the pressure will increase then the equilibrium will shift towards the fewer and then more number of the gas moles and hence, will have no net effect.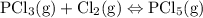 when the volume is increased then the pressure will decrease and the reaction will shift towards more moles of the gas present that is the left side.
when the volume is increased then the pressure will decrease and the reaction will shift towards more moles of the gas present that is the left side.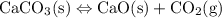 when the volume is increased then the pressure will decrease and the equilibrium will shift towards the side where more moles are present. Hence the increase in the volume will shift the equilibrium towards the right.
when the volume is increased then the pressure will decrease and the equilibrium will shift towards the side where more moles are present. Hence the increase in the volume will shift the equilibrium towards the right.

