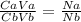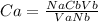Chemistry, 26.06.2019 11:00 anyaimartinez1901
25.0 ml of 0.212 m naoh is neutralized by 13.6 ml of an hcl solution. the molarity of the naoh solution is

Answers: 2


Another question on Chemistry


Chemistry, 23.06.2019 05:50
Which of the following isotopes has the same number of neutrons as phosphorus-31?
Answers: 1

Chemistry, 23.06.2019 07:00
0.88 moles of n2o5 (g) was placed in a sealed 1.00 l vessel. calculate the equilibrium concentration of n2o5. no2, and o2 and the equilibrium constant after equilibrium has been reached by 65.0% of the n2o5 decomposing.
Answers: 1

Chemistry, 23.06.2019 10:30
4al + 3o2 → 2al2o3 what does the "3" in front of o2 stand for? a) it indicates that there are 5 oxygen atoms after you add the coefficient and the subscript. b) it indicates that that there are are total of 6 oxygen atoms all bonded together as a single molecule. c) it indicates that there are 3 oxygen molecules chemically bonded to each other in the reaction. d) it indicates that there are 3 separate oxygen molecules in the reaction.
Answers: 2
You know the right answer?
25.0 ml of 0.212 m naoh is neutralized by 13.6 ml of an hcl solution. the molarity of the naoh solut...
Questions

English, 03.04.2020 06:58



Mathematics, 03.04.2020 06:58

Chemistry, 03.04.2020 06:58

Mathematics, 03.04.2020 06:58


Mathematics, 03.04.2020 06:58

Mathematics, 03.04.2020 06:58


Mathematics, 03.04.2020 06:58



Mathematics, 03.04.2020 06:58





English, 03.04.2020 06:58