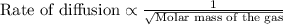Chemistry, 02.02.2020 18:42 xxxharveyweinsteinxx
the molar masses of four unknown gases are shown in the table.
molar mass of unknown gases
gas molar mass
a 44g/mol
b 20g/mol
c 30g/mol
d 32g/mol
which gas is likely to have the highest rate of effusion?
gas a
gas b
gas c
gas d

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 18:20
Complete the table for ion charge based upon their losing or gaining electrons in the outer shell. (use the periodic table as necessary.) group most likely ionic charge # of valence electrons i +1 ii +2 iii +3 iv +4 or -4 v -3 vi -2 vii -1 viii 0
Answers: 2

Chemistry, 21.06.2019 23:00
Plz choose one of the compounds from the table and explain how you know the numbers of atoms in your formula. is it possible for two different compounds to be made from the exact same two elements? why or why not? with a limited number of elements (less than 120 are known), does this mean we also have a small number of compounds or do we have a large number of compounds in this world?
Answers: 1

Chemistry, 22.06.2019 08:30
The mass of a neutron is equal to the mass of a proton plus the mass of an electron. true or false false true
Answers: 1

Chemistry, 22.06.2019 18:50
Question 3(multiple choice worth 4 points) (04.04 lc) what does it mean when an element is reduced? it empties a valance shell, reducing its atomic radius. it gains electrons, reducing its overall charge. it increases electronegativity, reducing its ability to bond. it loses electrons, reducing its electron number.
Answers: 1
You know the right answer?
the molar masses of four unknown gases are shown in the table.
molar mass of unknown gas...
molar mass of unknown gas...
Questions

Mathematics, 12.12.2019 21:31





Social Studies, 12.12.2019 21:31



Mathematics, 12.12.2019 21:31

Social Studies, 12.12.2019 21:31




Chemistry, 12.12.2019 21:31