The pH of the solution is 4.5
Explanation:
pH is defined as the negative logarithm of hydrogen or hydronium ion concentration. It ranges from the scale of 1 to 14.
The formula used to calculate pH of the solution is:
![pH=-\log[H_3O^+]](/tpl/images/0492/8473/a23b5.png)
We are given:
![[H_3O^+]=3.2\times 10^{-5}M](/tpl/images/0492/8473/e30f6.png)
Putting values in above equation, we get:
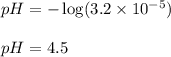
Hence, the pH of the solution is 4.5



























