
Chemistry, 06.12.2019 05:31 taylortayshaun7
The radioactive decay of a certain sample produced 846 disintegrations per minute. exactly 3.00 days later, the rate of decay was found to be 269 disintegrations per minute. calculate the half-life, in days, for the decay of this sample.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:30
How many moles of carbon dioxide will form if 2.5 moles of c3h8 is burned
Answers: 1

Chemistry, 22.06.2019 11:50
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2

Chemistry, 22.06.2019 12:10
|using the periodic tablewarm-upuse the periodic table in the tools bar to answer the following questions.what elemental classification does oxygen belongto? done
Answers: 3

Chemistry, 22.06.2019 21:30
What is another way to determine mass times acceleration?
Answers: 1
You know the right answer?
The radioactive decay of a certain sample produced 846 disintegrations per minute. exactly 3.00 days...
Questions




Physics, 21.07.2019 21:30


History, 21.07.2019 21:30





Biology, 21.07.2019 21:30


Biology, 21.07.2019 21:30


Chemistry, 21.07.2019 21:30


Social Studies, 21.07.2019 21:30


Mathematics, 21.07.2019 21:30

History, 21.07.2019 21:30

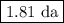
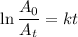
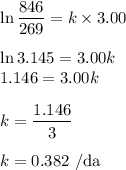
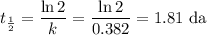
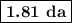 .
.

