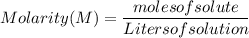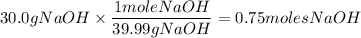Chemistry, 25.09.2019 16:30 ariellencevallo
What is the molarity of a solution prepared by dissolving 30.0 grams of naoh in enough water to make a solution with a total volume of 2.40 liters?
0.150 m naoh
0.218 m naoh
0.313 m naoh
0.462 m naoh

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:00
Water's surface tension and heat storage capacity are accounted for by its a) orbitals b) weight c) hydrogen bonds d) mass e) size
Answers: 2

Chemistry, 22.06.2019 12:00
Ageochemist examines a piece of metal that he found in the soil. he performs tests to identify the metal from its density, electrical conductivity, and melting point. which statement best describes his investigation? a. he is determining physical properties that are sufficient to identify the metal.b. he is determining chemical properties that are sufficient to identify the metal.c. he is determining physical properties that are insufficient to identify the metal.d. he is determining chemical properties that are insufficient to identify the metal.
Answers: 3


Chemistry, 22.06.2019 19:00
How does a catalyst increase the speed of a reaction? a. the catalyst eliminates the activated complex stage, allowing products to form immediately. b. the catalyst lowers the energy level of the reactants, making it easier for them to react. c. the catalyst makes it easier for the activated complex to form, lowering the activation energy. d. the catalyst raises the energy level of the products, making the reaction finish sooner. reset next
Answers: 1
You know the right answer?
What is the molarity of a solution prepared by dissolving 30.0 grams of naoh in enough water to make...
Questions

Mathematics, 13.07.2020 20:01


Mathematics, 13.07.2020 20:01


Mathematics, 13.07.2020 20:01


Geography, 13.07.2020 20:01

Mathematics, 13.07.2020 20:01


Mathematics, 13.07.2020 20:01


Mathematics, 13.07.2020 20:01

Mathematics, 13.07.2020 20:01


Social Studies, 13.07.2020 20:01

Mathematics, 13.07.2020 20:01

Health, 13.07.2020 20:01

Biology, 13.07.2020 20:01