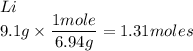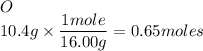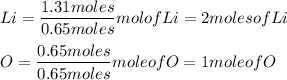Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 14:50
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3

Chemistry, 22.06.2019 15:30
The reactions of photosynthesis occur in the of plant cell? a.mitochondria. b. lysosomes. c. chloroplasts. d. chlorophyll
Answers: 1

Chemistry, 22.06.2019 22:00
How many moles of no2 will form when 3.3 moles of cu are reacted with excess hno3?
Answers: 3
You know the right answer?
Calculate the empirical formula 9.1 g of lithium and 10.4 g of oxygen...
Questions

Mathematics, 11.02.2022 23:40

English, 11.02.2022 23:40

History, 11.02.2022 23:40




History, 11.02.2022 23:50



Mathematics, 11.02.2022 23:50

Chemistry, 11.02.2022 23:50

History, 11.02.2022 23:50


History, 11.02.2022 23:50





Social Studies, 11.02.2022 23:50

Mathematics, 11.02.2022 23:50