
Chemistry, 22.12.2019 12:31 icecreamisgood4u
One of the main components of pearls is calcium carbonate (caco3). if pearls are put into an acidic solution, they dissolve: caco3(s) + 2hcl(aq) ➡ cacl2(aq) + h2o(l) + co2(g). how many moles of caco3 can be dissolved in 0.0250 moles of hydrochloric acid?
0.0125 moles
0.0500 moles
2.50 moles
4.50 moles

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:30
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

Chemistry, 22.06.2019 09:00
Look at the spectrums of a star moving towards earth and a motionless star. which of these is a correct inference that can be draw from the observation of the two spectrums? (2 points) the spectrum of a motionless star is difficult to be viewed separately using oridinary telescopes. the spectrum of a motionless star is identical to the spectrum of a star which moves towards earth. the spectrum of a star shifts towards the red region when the star moves towards earth. the spectrum of a star shifts towards the blue region when the star moves towards earth.
Answers: 2

Chemistry, 22.06.2019 15:20
An alloy contains 66 g of pure zinc. what is the percentage of zinc in the alloy? express your answer to two significant figures and include the appropriate units.
Answers: 1

You know the right answer?
One of the main components of pearls is calcium carbonate (caco3). if pearls are put into an acidic...
Questions



Arts, 19.07.2019 19:40

History, 19.07.2019 19:40


Biology, 19.07.2019 19:40


Physics, 19.07.2019 19:40



Chemistry, 19.07.2019 19:40

Physics, 19.07.2019 19:40

Chemistry, 19.07.2019 19:40

Physics, 19.07.2019 19:40

Social Studies, 19.07.2019 19:40

Computers and Technology, 19.07.2019 19:40

English, 19.07.2019 19:40

Chemistry, 19.07.2019 19:40

World Languages, 19.07.2019 19:40

Biology, 19.07.2019 19:40

![CaCO_3(s)+2HCl(aq)]\rightarrow CaCl_2(aq)+H_2O(l)+CO_2(g)](/tpl/images/0429/7730/2251f.png)
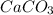
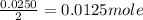 of
of 

