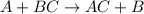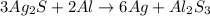In the presence of hydrogen sulfide, silver oxidizes to form silver sulfide (silver tarnish). it forms a black film on the surface of silver. 4ag 2h2s o2→2ag2s 2h2o one way to reverse the chemical reaction and turn silver sulfide back into silver is by immersing the silver in an aluminum container filled with a hot solution of sodium bicarbonate. metals like aluminum have a greater affinity for sulfur than silver. this tarnish-removal method uses a chemical reaction to convert the silver sulfide back into silver, resulting in no net loss of silver. the reaction between silver sulfide and aluminum takes place in the presence of a sodium bicarbonate (nahco3) solution, resulting in the transfer of sulfur atoms from silver to aluminum, freeing the silver metal and forming aluminum sulfide. the reaction can be summarized as follows: when silver sulfide reacts with aluminum, silver sulfide is reduced to form silver and aluminum is oxidized to form aluminum sulfide. complete the balanced chemical equation for the reaction, showing the formation of silver metal from silver sulfide in the presence of aluminum metal. express your answer as the products of a chemical equation. view available hint(s) 3ag2s 2al → 3 a g 2 s 2 a l →

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:00
Which best describes how johannes kepler developed his laws of planetary motion
Answers: 3

Chemistry, 22.06.2019 04:30
Turbo the snail moves across the ground at a pace of 12 feet per day. if the garden is 48 feet away, how many days will it take for the snail to get there?
Answers: 2

Chemistry, 22.06.2019 06:00
Calculate - analysis of compound composed of iron and oxygen yields 174.86 of fe and 75.14g of o. what is the empirical formula for this compound?
Answers: 3

You know the right answer?
In the presence of hydrogen sulfide, silver oxidizes to form silver sulfide (silver tarnish). it for...
Questions


Mathematics, 24.04.2020 21:48



Health, 24.04.2020 21:48





World Languages, 24.04.2020 21:48





Mathematics, 24.04.2020 21:49




English, 24.04.2020 21:49