Chemistry, 29.01.2020 17:48 Delgadojacky0206
[standard enthalpy of formation]
remember to show work and explain.
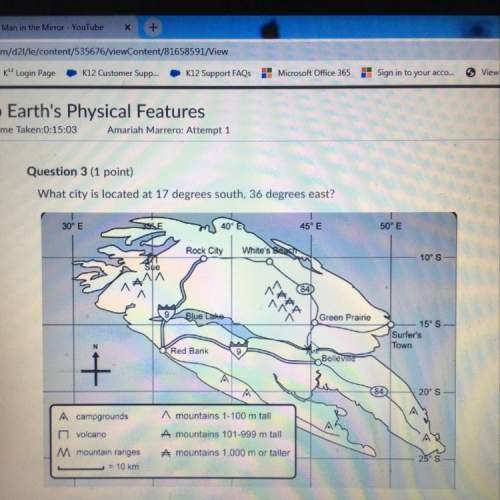
5. calculate the change in enthalpy for:
6. calculate the ∆h_rxn given the following
![[standard enthalpy of formation]remember to show work and explain.5. calcula](/tpl/images/00/29/OYuilGD6V9dfCUtT.jpg)

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 21:30
Sex cells from female reproductive organ? 1) mitosis 2) fertilization 3) zygote 4) eggs 5) meiosis 6) sperm
Answers: 2

Chemistry, 22.06.2019 20:00
Carbon-14 undergoes radioactive decay in the reaction above. determine the type of radiation emitted in this reaction and describe what is happening to the nucleus during this reaction.
Answers: 2


Chemistry, 23.06.2019 07:00
If you used the method of initial rates to obtain the order for no2, predict what reaction rates you would measure in the beginning of the reaction for initial concentrations of 0.200 m, 0.100 m, & 0.050 m no2.
Answers: 3
You know the right answer?
[standard enthalpy of formation]
remember to show work and explain.
5. calcula...
remember to show work and explain.
5. calcula...
Questions

Mathematics, 16.03.2020 21:08

English, 16.03.2020 21:08













Computers and Technology, 16.03.2020 21:08




Computers and Technology, 16.03.2020 21:08

Mathematics, 16.03.2020 21:08