
Which of the following electron configurations is most likely to result in an element that is relatively inactive?
a. a filled energy sublevel
b. a half-filled energy sublevel
c. one empty and one filled energy sublevel
d. a filled highest occupied principal energy level

Answers: 1


Another question on Chemistry

Chemistry, 23.06.2019 04:31
What are the coefficients that will balance the skeleton equation below? n2 + h2 → nh3
Answers: 1

Chemistry, 23.06.2019 08:30
Of the following elements, which is the least reactive? a. c b. h c. li d. he
Answers: 1

Chemistry, 23.06.2019 09:20
Description: biological systems 5 differ from chemical systems when it comes to equilibrium. in a chemical system once equilibrium is met, no other reactions occur. in a biological system, a dynamic equilibrium is used when a substrate is turned into a product, another reaction creates the same substrate thus keeping the concentrations stagnant. this allows for cells to continually make new compounds without messing the delta g for the systems instructions: write a response to the following prompt and then respond to your peers. your individual response is due thursday at midnight (cst) your response to your peers is due saturday at midnight (cst) prompt: propose what would happen if a living cell all the sudden reached chemical equilibrium. also discuss the effects of build up of a particular substrate on a biological system. how would this affect overall delta g's?
Answers: 3

Chemistry, 23.06.2019 11:30
How many grams of carbon are in 237 grams of ethanol(c2h5oh) and how many sulfide ions are in 2.45 moles of aluminum sulfide show me you you got the answers
Answers: 3
You know the right answer?
Which of the following electron configurations is most likely to result in an element that is relati...
Questions

Mathematics, 18.03.2021 01:40



Mathematics, 18.03.2021 01:40


Spanish, 18.03.2021 01:40

Geography, 18.03.2021 01:40



Mathematics, 18.03.2021 01:40

Computers and Technology, 18.03.2021 01:40



Mathematics, 18.03.2021 01:40


Mathematics, 18.03.2021 01:40

Chemistry, 18.03.2021 01:40



Mathematics, 18.03.2021 01:40

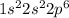 .
.

