
Chemistry, 30.01.2020 10:56 KENYONWASHINGTON868
How many moles of zncl2 will be produced from 61.0g of zn, assuming hcl is available in excess?
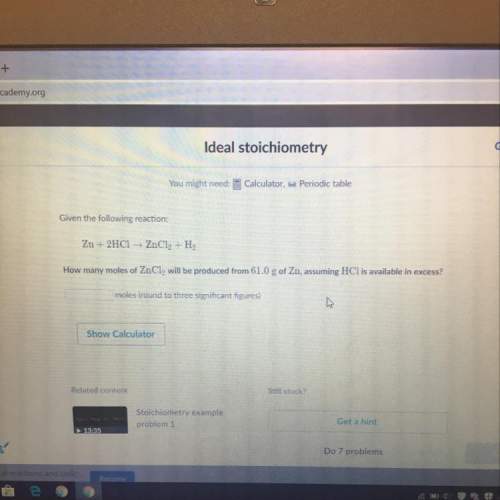

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:30
This chart represents the melting point of several substance. what besy explains the high melting point of the salt?
Answers: 2

Chemistry, 22.06.2019 04:50
Acompound contains c, h, and o atoms. when 1.130 g of the compound is burned in oxygen, 1.064 g co2 and 0.3631 g h2o are produced. what is the empirical formula of this compound?
Answers: 1

Chemistry, 22.06.2019 05:30
What type of reaction is shown below? check all that apply. 2h2o2 → 2h2o + o2 synthesis decomposition combustion
Answers: 1

Chemistry, 22.06.2019 09:00
Plz mark brainliest 30 points1) find the momentum of a 12 kg snowball that is rolling with a velocity of 9 m/s.2) an 8 ball with a mass of .5 kg is sitting at rest. it is hit by the cue ball (1 kg) traveling at 2.5 m/s. if the cue ball is at rest after the collision, how fast is the 8 ball traveling after the collision? 3) two football players are running toward each other. if the offensive player is 75 kg and is running 8 m/s, how fast must the 60 kg defensive player run in order for the two players to hit and stop?
Answers: 1
You know the right answer?
How many moles of zncl2 will be produced from 61.0g of zn, assuming hcl is available in excess?
Questions


Mathematics, 27.05.2021 01:00

Mathematics, 27.05.2021 01:00



Mathematics, 27.05.2021 01:00

Mathematics, 27.05.2021 01:00


English, 27.05.2021 01:00

Mathematics, 27.05.2021 01:00


History, 27.05.2021 01:00

Biology, 27.05.2021 01:00


Mathematics, 27.05.2021 01:00




Mathematics, 27.05.2021 01:00

Mathematics, 27.05.2021 01:00



