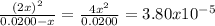The dissociation of molecular iodine into iodine atoms is represented as i2(g) ⇌ 2i(g) at 1000 k, the equilibrium constant kc for the reaction is 3.80 × 10−5. suppose you start with 0.0456 mol of i2 in a 2.28−l flask at 1000 k. what are the concentrations of the gases at equilibrium? what is the equilibrium concentration of i2

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 07:00
How heavy is thanos? a) 3000 lbs b) all of it c) the price of tea in china d) heavy enough
Answers: 2


Chemistry, 22.06.2019 14:30
Need ! asap will mark 10 pts using the room temperature line (orange line) and your periodic table, make lists that identify the state of matter (gas, liquid, or solid) in which each element you plotted exists at room temperature. explain your answers
Answers: 1

Chemistry, 22.06.2019 23:30
The density of benzene at 15 °c is 0.8787 g/ml. calculate the mass of 0.1500 l of benzene at this temperature. enter your answer in terms of grams
Answers: 2
You know the right answer?
The dissociation of molecular iodine into iodine atoms is represented as i2(g) ⇌ 2i(g) at 1000 k, th...
Questions



Mathematics, 26.08.2019 06:00


Business, 26.08.2019 06:00

Mathematics, 26.08.2019 06:00




Chemistry, 26.08.2019 06:00

English, 26.08.2019 06:00

Social Studies, 26.08.2019 06:00

Geography, 26.08.2019 06:00

Mathematics, 26.08.2019 06:00

Mathematics, 26.08.2019 06:00

Mathematics, 26.08.2019 06:00




![\frac{[I]^{2} }{[I_{2} ]}](/tpl/images/0472/7611/f811c.png)
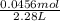 → [I₂]₀ = 0.0200 M
→ [I₂]₀ = 0.0200 M![\frac{[I]^{2} }{[I_{2} ]} = \frac{(2x)^{2} }{0.0200 - x} = 3.80x10^{-5}](/tpl/images/0472/7611/3afcc.png)