Chemistry, 03.02.2020 05:42 ketnerfaith17
The methyl red solution you will use for this experiment was made using .080 grams of methyl red per liter of solution. the molar mass of methyl red is 269.31 g/mole. what is the molarity of the stock solution you will be using?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 18:00
Balance this equation: n2 + h2 > nh3, write the following molar ratios: n2 / n2 / nh3 h2 /
Answers: 1


Chemistry, 22.06.2019 06:40
Which statement is usually true about the relationship between activation energy and reaction rates? low activation energy barriers result in low rates. high activation energy barriers result in low rates. low activation energy barriers result in no reaction. high activation energy barriers result in no reaction.
Answers: 3

Chemistry, 22.06.2019 12:00
Give the set of reactants (including an alkyl halide and a nucleophile) that could be used to synthesize the following ether: draw the molecules on the canvas by choosing buttons from the tools (for bonds and charges), atoms, and templates toolbars, including charges where needed. ch3ch2och2ch2chch3 | ch3
Answers: 1
You know the right answer?
The methyl red solution you will use for this experiment was made using .080 grams of methyl red per...
Questions




















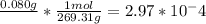 moles
moles