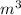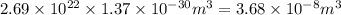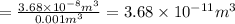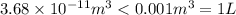Chemistry, 30.01.2020 04:42 FIRENINJA117
Asample of gaseous neon atoms at atmospheric pressure and 0 °c contains 2.69 * 1022 atoms per liter. the atomic radius of neon is 69 pm. what fraction of the space do the atoms occupy? what does this reveal about the separation between atoms in the gaseous phase?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 04:30
There is a single path for electrons. the current decreases when additional resistors are added. the current will be the same in each resistor. these statements best describe a(n) circuit.
Answers: 3

Chemistry, 22.06.2019 13:30
Mary is conducting an experiment on how pollution affects plant growth. how can she ensure that her data are reliable?
Answers: 3

Chemistry, 22.06.2019 13:30
Some animals that try to adapt to climate changes eventually die due to starvation, as climate change alters the web.
Answers: 2

Chemistry, 22.06.2019 17:10
In which block of the periodic table is uranium (u) found? s blockd blockp blockf block
Answers: 1
You know the right answer?
Asample of gaseous neon atoms at atmospheric pressure and 0 °c contains 2.69 * 1022 atoms per liter....
Questions



History, 30.03.2020 22:29

Mathematics, 30.03.2020 22:29

Social Studies, 30.03.2020 22:29



Chemistry, 30.03.2020 22:29


Mathematics, 30.03.2020 22:29


Mathematics, 30.03.2020 22:29





Mathematics, 30.03.2020 22:29



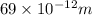
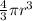
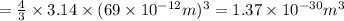
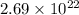 atoms are present in 1L
atoms are present in 1L